The main aim of the TROG Scientific Committee (TSC) is to ensure that TROG’s mission; to conduct world-class research in radiation medicine that leads the global effort to better control and cure cancer and to help improve outcomes for people affected by cancer is met.
TROG aims to conduct high quality, practice changing clinical trials, the TSC supports this through facilitated discussion and endorsement of clinical trial concepts and proposals submitted by our members via the TROG New Proposals Pathway, oversight of active trials and ensuring publication of trial results.
The TSC is composed of the members listed below, who are experts in the fields of radiation oncology, medical oncology, interventional oncology, medical physics, clinical research, quality of life research, health economics, statistics, and trial coordination.
Scientific Committee Chair
Scientific Committee Deputy Chair
Radiation Oncologist
Radiation Oncologist
Radiation Oncologist
Discipline Representative: Medical Oncology
Discipline Representative: Interventional Oncology
Statistics
Health Economist
QoL Technical Service
Radiation Therapist
Independent Consumer Representative
TROG Research Manager
TROG Radiation Therapy Manager
Secretary
TROG and the TSC work closely with our members to ensure that our trials and new proposals address the key priorities in radiation oncology and that we are horizon scanning for emerging technologies and techniques in radiation medicine.
A number of working parties, committees and special interest groups provide guidance to the TSC including:
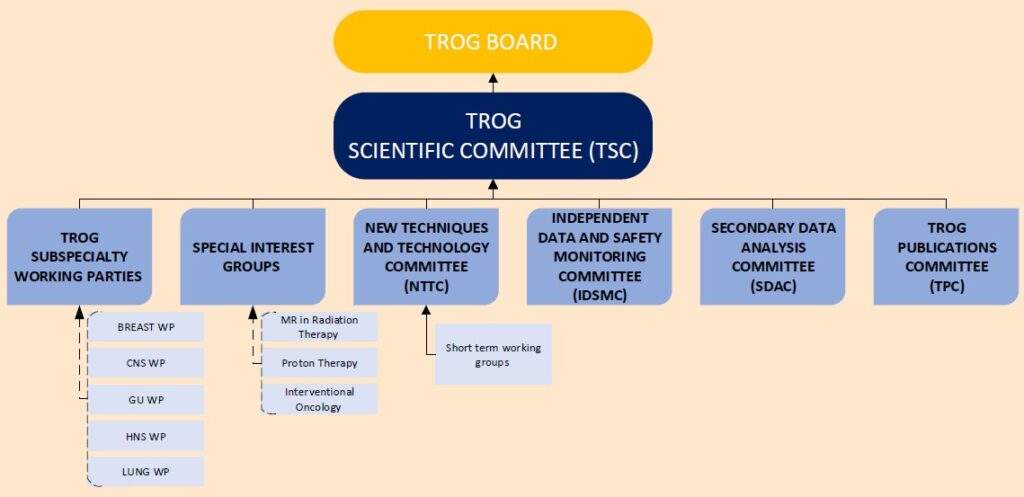
The participation of our membership in subspecialty working parties, committees and special interest groups and brings new ideas, partnerships, collaborations, guidelines, and policies that support advancing radiation medicine research to improve outcomes for those affected by cancer.
Contact the TROG team at committees@trog.com.au for further information.
For over 30 years, Trans-Tasman Radiation Oncology Group has been dedicated to improving the way radiation medicine is delivered to cancer patients with ongoing scientific research, clinical trials, and cutting-edge technology.
Latest News
Keep up to date with TROG news, subscribe to our community newsletter.
ABN: 45 132 672 292